Burkholderia pseudomallei (also known as Pseudomonas pseudomallei) is a Gram-negative, bipolar, aerobic, motile rod-shaped bacterium. It infects humans and animals and causes the disease melioidosis. It is also capable of infecting plants.
B. pseudomallei measures 2-5 μm in length and 0.4-0.8 μm in diameter and is capable of self-propulsion using flagellae. The bacteria can grow in a number of artificial nutrient environments, especially betaine- and arginine-containing ones.
In vitro, optimal proliferation temperature is reported around 40°C in neutral or slightly acidic environments (pH 6.8â€"7.0). The majority of strains are capable of fermentation of sugars without gas formation (most importantly, glucose and galactose, older cultures are reported to also metabolize maltose and starch). Bacteria produce both exo- and endotoxins. The role of the toxins identified in the process of melioidosis symptom development has not been fully elucidated.
Identification
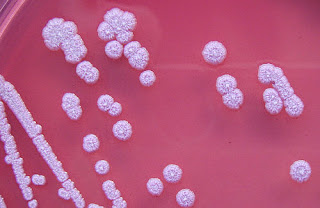
B. pseudomallei is not fastidious and will grow on a large variety of culture media (blood agar, MacConkey agar, EMB, etc.). Ashdown's medium (or Burkholderia cepacia medium) may be used for selective isolation. Cultures typically become positive in 24 to 48 hours (this rapid growth rate differentiates the organism from B. mallei, which typically takes a minimum of 72 hours to grow). Colonies are wrinkled, have a metallic appearance, and possess an earthy odour. On Gram staining, the organism is a Gram-negative rod with a characteristic "safety pin" appearance (bipolar staining). On sensitivity testing, the organism appears highly resistant (it is innately resistant to a large number of antibiotics including colistin and gentamicin) and that again differentiates it from B. mallei, which is in contrast, exquisitely sensitive to a large number of antibiotics. For environmental specimens only, differentiation from the nonpathogenic B. thailandensis using an arabinose test is necessary (B. thailandensis is never isolated from clinical specimens). The laboratory identification of B. pseudomallei has been described in the literature.
The classic textbook description of B. pseudomallei in clinical samples is of an intracellular, bipolar-staining, Gram-negative rod, but this is of little value in identifying the organism from clinical samples. Some suggest the Wayson stain is useful for this purpose, but this has been shown not to be the case.
Laboratory identification of B. pseudomallei can be difficult, especially in Western countries where it is rarely seen. The large wrinkled colonies look like environmental contaminants, so are often discarded as being of no clinical significance. Colony morphology is very variable and a single strain may display up multiple colony types, so inexperienced laboratory staff may mistakenly believe the growth is not pure. The organism grows more slowly than other bacteria that may be present in clinical specimens, and in specimens from nonsterile sites, is easily overgrown. Nonsterile specimens should, therefore, be cultured in selective media (e.g., Ashdown's or B. cepacia medium). For heavily contaminated samples, such as faeces, a modified version of Ashdown's that includes norfloxacin, amoxicillin, and polymyxin B has been proposed.
Even when the isolate is recognised to be significant, commonly used identification systems may misidentify the organism as Chromobacterium violaceum or other nonfermenting, Gram-negative bacilli such as Burkholderia cepacia or Pseudomonas aeruginosa. Again, because the disease is rarely seen in western countries, identification of B. pseudomallei in cultures may not actually trigger alarms in physicians unfamiliar with the disease. Routine biochemical methods for identification of bacteria vary widely in their identification of this organism: the API 20NE system accurately identifies B. pseudomallei in 99% of cases, as does the automated Vitek 1 system, but the automated Vitek 2 system only identifies 19% of isolates.
The pattern of resistance to antimicrobials is distinctive, and helps to differentiate the organism from P. aeruginosa. The majority of B. pseudomallei isolates are intrinsically resistant to all aminoglycosides (via an efflux pump mechanism), but sensitive to co-amoxiclav: this pattern of resistance almost never occurs in P. aeruginosa and is helpful in identification. Unfortunately, the majority of strains in Sarawak, Borneo, are susceptible to aminoglycosides and macrolides, which means the conventional recommendations for isolation and identification do not apply there.
Molecular methods (PCR) of diagnosis are possible, but not routinely available for clinical diagnosis. Fluorescence in situ hybridisation has also been described, but has not been clinically validated, and it not commercially available . In Thailand, a latex agglutination assay is widely used, while a rapid immunofluorescence technique is also available in a small number of centres.
Disinfection
B. pseudomallei is susceptible to numerous disinfectants, including benzalkonium chloride, iodine, mercuric chloride, potassium permanganate, 1% sodium hypochlorite, 70% ethanol, 2% glutaraldehyde, and to a lesser extent, phenolic preparations. B. pseudomallei is effectively killed by the commercial disinfectants, Perasafe and Virkon. The microorganism can also be destroyed by heating to above 74°C for 10 min or by ultraviolet irradiation. B. pseudomallei is not reliably disinfected by chlorine.
Medical importance
B. pseudomallei infection in humans is called melioidosis. The mortality of melioidosis is 20 to 50% even with treatment.
Antibiotic treatment and sensitivity testing
The antibiotic of choice is ceftazidime. While various antibiotics are active in vitro (e.g., chloramphenicol, doxycycline, co-trimoxazole), they have been proven to be inferior in vivo for the treatment of acute melioidosis. Disc diffusion tests are unreliable when looking for co-trimoxazole resistance in B. pseudomallei (they greatly overestimate resistance) and Etests or agar dilution tests should be used in preference. The actions of co-trimoxazole and doxycycline are antagonistic, which suggests these two drugs ought not to be used together.
The organism is intrinsically resistant to gentamicin and colistin, and this fact is helpful in the identification of the organism. Kanamycin is used to kill B. pseudomallei in the laboratory, but the concentrations used are much higher than those achievable in humans.
Pathogenicity mechanisms and virulence factors
B. pseudomallei is an "accidental pathogen". An environmental organism, it has no requirement to pass through an animal host to replicate. From the point of view of the bacterium, human infection is an evolutionary "dead end".
Strains which cause disease in humans differ from those causing disease in other animals by possessing certain genomic islands. It may have the ability to cause disease in humans because of DNA it has acquired from other microorganisms. The mutation rate is also high, and the organism continues to evolve even after infecting the host.
B. pseudomallei is able to invade cells (it is an intracellular pathogen). It is able to polymerise actin and to spread from cell to cell, causing cell fusion and the formation of multinucleated giant cells. The bacterium also expresses a toxin called lethal factor 1. B. pseudomallei is one of the first Proteobacteria to be identified as containing an active type-6 secretion system. it is also the only organism identified that contains up to six different ones.
B. pseudomallei is intrinsically resistant to a large number of antimicrobial agents. One important mechanism is that it is able to pump drugs out of the cell, and this mediates resistance to aminoglycosides (AmrAB-OprA), tetracyclines, fluoroquinolones, and macrolides (BpeAB-OprB).
Vaccine candidates
No vaccine is currently available, but a number of vaccine candidates have been suggested. Aspartate-β-semialdehyde dehydrogenase (asd) gene deletion mutants are auxotrophic for diaminopimelate (DAP) in rich media and auxotrophic for DAP, lysine, methionine, and threonine in minimal media. The Î"asd bacterium (bacterium with the asd gene removed) protects against inhalational melioidosis in mice.
References
External links
- Burkholderia pseudomallei genomes and related information at PATRIC, a Bioinformatics Resource Center funded by NIAID
- Getting a Grip on the Great Mimicker: Secrets of a Stealth Organism from the Wellcome Trust.
- Pathema Burkholderia resource